Insights on Covalent Attachment of a Re Bipyridyl Catalyst onto p-Si(111) Using an Alkyl Linker for Photoelectrochemical CO2 Reduction #474
Authors
Byunghoon Lee, Christopher J. Miller, Jessica G. Freeze, Rajiv Ramanujam Prabhakar, Glenda Chen, Saya Okuno, Julianne S. Lampert, Victor S. Batista, Clifford P. Kubiak
Abstract
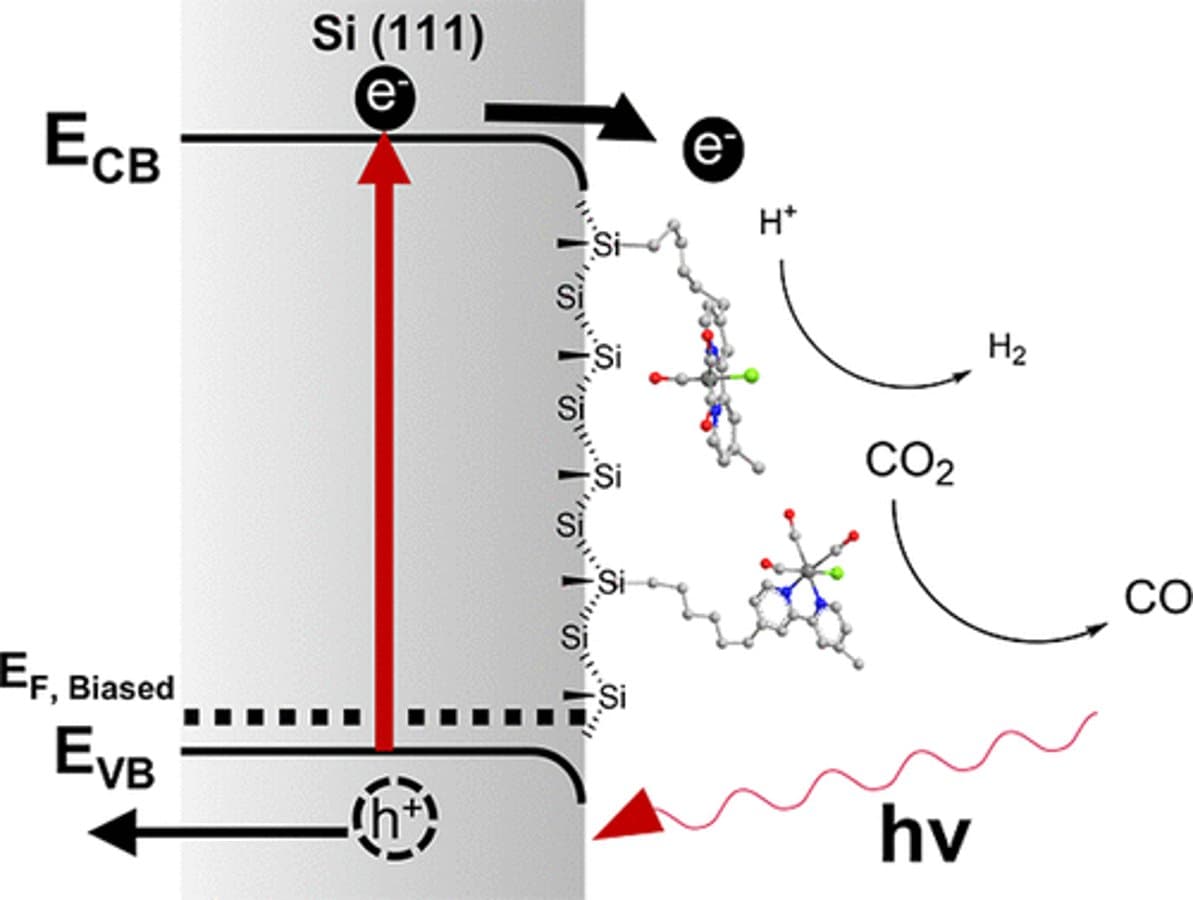
Re(bpy)(CO)3Cl with a hexyl chain linker was covalently attached to p-type Si(111) via Si–C bond using an organolithium route and confirmed by XPS and ATR-FTIR. Cyclic voltammetry (CV) shows two single-electron reductions with estimated photovoltages of 320 mV and 400 mV for the first and second reductions, respectively. Both reductions were shifted by approximately 200 mV to more positive potentials relative to the homogeneous analogue measured with a methyl-terminated p-type Si(111). CV in a concentrated Cl– solution indicated that the reformation of the Re–Cl bond is slow, indicating a kinetic barrier in reactivity at the active site. DFT geometry optimizations suggest that under negative bias the complex preferentially adopts configurations that move farther from the methylated Si surface, with the Re–Cl bond oriented parallel to it. Under CO2, only slight catalytic current enhancement was observed. Controlled-potential electrolysis (CPE) at −2.3 V vs Fc+/0 yielded Faradaic efficiencies (FEs) of 15% for CO and 54% for H2, compared to ∼100% FE for CO observed in the homogeneous system. The selectivity shift is best explained by the dominance of HER at the native Si surface, while CO formation remains limited by slow EC kinetics at the active site of the immobilized Re complex.