Electrochemical Nitrate Reduction to Ammonia Driven by Catalytic Monovacancies in Single-Walled Carbon Nanotubes #470
Authors
Jana Jelušić, Nia J Harmon, Jan Paul Menzel, Shize Yang, Quentin C Bertrand, Hailiang Wang, Gary W Brudvig, Victor S Batista
Abstract
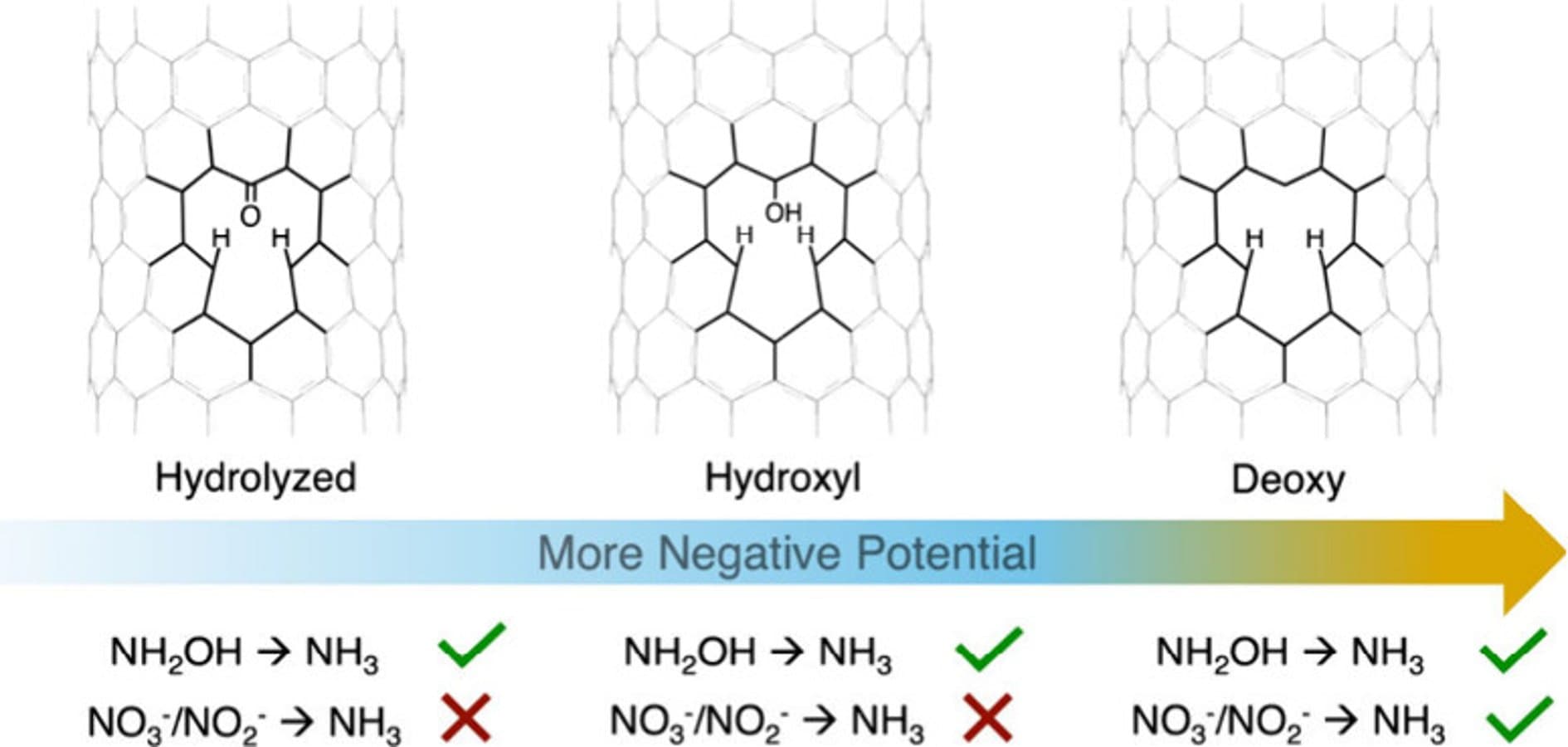
Developing alternative routes for ammonia (NH3) synthesis from nitrogen-containing species under mild conditions is a central challenge in sustainable catalysis. Single-walled carbon nanotubes (SWCNTs) containing intrinsic monovacancy defects provide a distinct class of active sites for electrochemical ammonia (NH3) production. Here, we investigate the reactivity of SWCNT monovacancies in the electrochemical reduction of nitrate (NO3–), nitrite (NO2–), and hydroxylamine (NH2OH) to NH3. We find that NO3– and NO2– reduction proceeds through a single proton-coupled electron transfer (PCET) pathway that requires regeneration of the vacancy site. In contrast, NH2OH reduction can occur through both vacancy-dependent and vacancy-independent mechanisms. At more negative potentials, NH2OH reacts at the regenerated vacancy to form either a ketone and NH3 or an oxime intermediate, which subsequently yields NH3 through additional PCET steps. These results establish SWCNT monovacancies as well-defined model systems for probing reaction mechanisms and guiding the design of efficient electrocatalysts for nitrate-to-ammonia conversion.